The CDC’s Advisory Committee on Immunization Practices (ACIP), voted unanimously Wednesday to add COVID-19 mRNA vaccinations to the Vaccines for Children program. The government must add these vaccines to the VFC program if a patient cannot afford them. This is to “ensure access.” In addition, vaccine manufacturers are exempt from liability for vaccine injuries since the product is on VFC. The government will continue to pay for vaccines and the manufacturers will be held responsible. This vote could be called the “Pfizer/Moderna continuing profit plan.”
CDC attempted to separate the VFC and the vaccination schedule during Wednesday’s discussion but its rhetoric was absurd. If ACIP does not place the vaccine on the schedule, no vaccine will be added to the VFC list. Why would the government pay for vaccines that aren’t recommended by ACIP?
The Wednesday vote set up performative voting on Thursday to include the COVID-19 vaccinations in the vaccine schedules for all Americans. Since December 2020 when the COVID-19 vaccines were launched, the CDC has been updating its recommendations. It has already been recommended that all Americans aged over six months receive mRNA vaccines.
The CDC actually issued an interim schedule for the COVID-19 vaccine on Monday for Americans aged over six months. ACIP recommended the ACIP interim vaccine schedules. ACIP recommended COVID boosters and vaccines for different populations. No one will be surprised to learn that ACIP voted again unanimously on Thursday to include them in the immunization schedules of children and adolescents.
The vaccine schedule is used by both state governments and physicians. It is used by doctors to recommend vaccines to patients. To create mandates for sports participation and school attendance, some states use the schedule. The immunization schedule of children aged 6 to 6 years was similar before the introduction of COVID vaccines.

The CDC will now add three doses to its schedule of an mRNA COVID vaccination for children younger than six years old, following Thursday’s vote. The appropriate vaccine schedule will include details on the dosage and timing of both COVID vaccine brand names, as well as information about different health statuses and age groups.
It is all semantics. The CDC recommendations are followed by most physicians. The vote updates the schedule for everyone. However, the only change after ACIP approves vaccine schedules is how many states automatically include the new vaccines in school attendance requirements and sports participation requirements.
An analysis by the University of Illinois Chicago found that 31 states and Washington D.C. have laws that require children to receive age-appropriate immunizations as recommended by ACIP. Some children also need boosters in the later grades, those marked with an ACIP triangle reference.
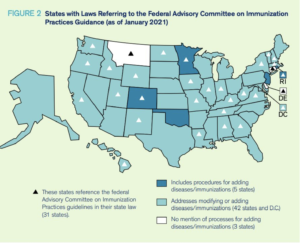
Seventeen states have laws that refer to the American Academy of Pediatrics (AAP), while ten state laws address FDA approval. Before ACIP added COVID-19 vaccines to the schedule, the AAP followed the CDC recommendations. All COVID-19 vaccinations that are currently being used in the United States have an emergency use authorization (EUA). The current Pfizer or Moderna vaccines are not FDA approved if a state law refers to FDA approval.
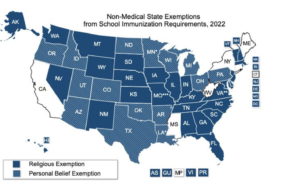
States have different ways of managing exemptions. Although all 50 states and Washington D.C. permit medical exemptions, the requirements for each state are different. California, Connecticut and Maine, Mississippi, New York, West Virginia, and New York allow medical exemptions. All other states and D.C. allow religious exemptions, with different requirements. 15 states offer exemptions based on personal beliefs, which gives parents more flexibility when it comes to vaccinating their children.
The ACIP recommendations may be broken if the COVID-19 vaccine is added to the child’s immunization schedule. After it was discovered that the mRNA vaccines were not effective in preventing illness and transmission, the CDC redefined what a vaccine is. Current products can also be offered under a EUA. Each state must decide whether it can accept ACIP recommendations under these extraordinary conditions.
Paul Offit, a member FDA’s vaccine advisory panel, stated that COVID vaccines are “in some ways, it is a matter of your style.” Some parents might argue that if there is any benefit, then they will accept the risk. Others may argue that there is no risk and they don’t want the risk if it’s low. It doesn’t seem to have much impact on otherwise healthy young people. Parents are increasingly seeking educational freedom. The demand for medical freedom by the same group of people should not be surprising to lawmakers.